
The n:p ratio increases, and the daughter nuclide lies closer to the band of stability than did the parent nuclide.Įlectron capture occurs when one of the inner electrons in an atom is captured by the atom’s nucleus. Positron decay is the conversion of a proton into a neutron with the emission of a positron. These nuclides lie below the band of stability. Electron capture occurs when one of the inner electrons in an atom is captured by the atom’s nucleus. 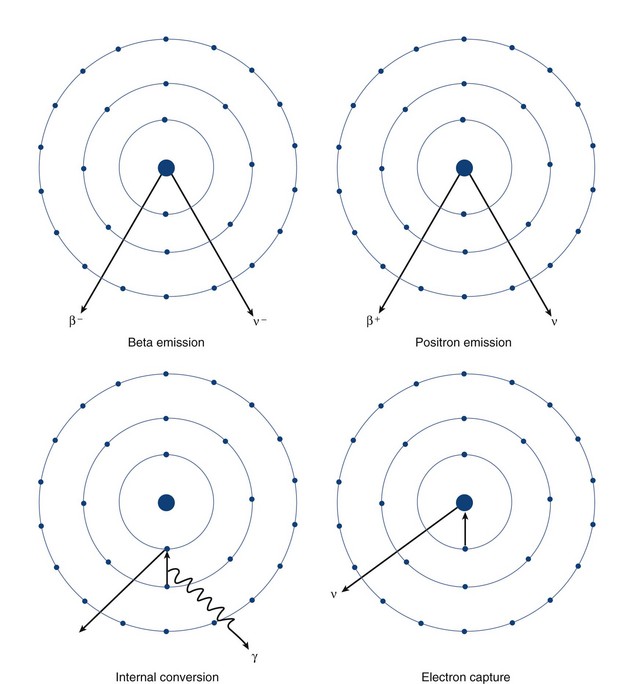
Mass number stays the same, Z decreases by 1. The n:p ratio increases, and the daughter nuclide lies closer to the band of stability than did the parent nuclide. Simplified for what you need to know for MCAT (AKA no neutrinos, etc.) Beta + decay: you emit a positron ( + ), in turn converting a proton to a neutron (think charge balance, you 'lost' and emitted the positive charge of the proton, in turn forming a neutron). Positron emission is observed for nuclides in which the n:p ratio is low. The radiation produced during radioactive decay is such that the daughter nuclide lies closer to the band of stability than the parent nuclide, so the location of a nuclide relative to the band of stability can serve as a guide to the kind of decay it will undergo ( Figure 21.5 ). Positron decay is the conversion of a proton into a neutron with the emission of a positron.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
